COSMOLOGY OF A BINARY
UNIVERSE
Part II
The Evolution of Physical
and Biological Complexity
( January 2008)
Preface: The remarkable ability of the Chaplygin/Tangent
gas, seen as a universal field (UF), to unify the forces of physics and to
offer an explanation for the observed accelerated expansion of the universe has
prompted further exploration of its possible use in problems of cosmic and
biological evolution.
In Part I, the
evolution of the UF itself was explored before and after the Big Bang
singularity. The unfolding of our atomic and molecular world of astronomy which
originated at the Big Bang was left for this Part II. However, this area has
two aspects, one is the expanding material cosmos of atoms and molecules since
the Big Bang singularity in which it originated, and the other is the development of the order and complex variety
within this material world, which we call evolution.
The expansion
phase of the atomic and molecular cosmos over the first 9 billion years or so
has, until recently, been dealt with in cosmology by the general theory of
relativity, which has had its difficulties.
These stem from the fact that general relativity (1) has as its basic
equation a postulated geometrical space and time (x,y,z,t) field or continuum
in which accelerations ( i.e. gravitational force) are the results of a
geometrical curvature of the postulated
continuum, (2) its dynamical equation of force is a differential equation with
no unique solution, and (3) it has no equation of state to physically
account for the expansion of the cosmos which it is used to describe.
The UF on the
contrary is a compressible physical field, whose basic equation is an integral
equation of state with a unique solution, and whose expansion is naturally
explained. Its force equation likewise stems from an integral energy equation whose space gradients set up
the forces, explains their existence in a physical, instead of a geometrical,
way, and unifies them all for the first time.
The world of
material molecules and atoms since the Big Bang can be envisaged as a
quasi-compressible fluid of atoms and molecules with an equation of state
relating pressure, specific volume and temperature. However, the observed
cosmos with its galaxies, clusters and immense voids is certainly not a close
approximation to an ideal expandable gas, so that this approach is a matter for
experts who have a full range of the essential data at hand. Consequently, for now, instead of treating
the cosmology of the post-Big Bang expansion, we shall turn to the development
of physical order and complexity and to biological evolution.
Although progress in the
formulation of a satisfactory theory of biological evolution has been
punctuated by a series of radical revisions, the thesis that biological
complexity has evolved appears to be well established.
Darwin’s original explanatory
theory, that this evolution has occurred by a natural selection of chance
biological variations passed from one generation to the next, suffered a check
when Mendel’s laws of inheritance were re-discovered. Mendel’s obstacle was the
fixed inheritance of genetic characteristics, which was further solidified with
the discovery of the genes and their DNA/RNA mechanisms. After a considerable
time, Darwinism was revived as neo-Darwinism when probabilistic laws of gene
combination were introduced, and the proposed evolutionary mechanism became, in
effect, gene emergence probability.
However, the basic physics or
biophysics of the problem still remained fundamentally unresolved, because the
emergence of complexity in nature, such as in the formation of genes, appears
to be in opposition to the 2nd law of thermodynamics. In spite of
this problem, plausible, but essentially incomplete, theories of automatic
emergence of physical complexity skirting the 2nd law have been
developed, extended to the emergence of the genes, and a near final solution to
the problem of the evolutionary mechanism is now sometimes asserted to have
been achieved.
Some serious attempts to
reconcile the emergence of complexity with the obstacle of the 2nd
law have been made, but these have not won general acceptance. In practice, the
2nd law problem is often just set aside as only a detail, destined to
inevitably be resolved in some manner yet unknown. Detailed analysis of the emergence of
physical complexity and its connection with evolutionary biology has been undertaken,
but these efforts have lacked any integration with the underlying orderly
physical forces that are the dynamism of change. The present situation might,
not too unkindly, be characterized as an optimistic expectation that all will
eventually be well and that, notwithstanding the 2nd law, biological order will
eventually somehow emerge from random physical disorder.
Now, in the last few years,
the emergence of the concept of a universal wave and force field (UF), derived
from the so-called Chaplygin/Tangent gas
of aeronautics and gas dynamics, has introduced a new level of basic physical
order. This approach has succeeded in
unifying the fundamental forces of physics, and so reconciles gravitation,
electromagnetism and quantum physics. Based on this standard dynamics and
thermodynamics of a compressible fluid medium, orderly force fields pose a
fundamental problem for the neo-Darwinian theoretical assertion of a purely
probabilistic emergence of physical complexity. This source of basic order in
both physics and biology, and its interaction with probability to produce the
complex physical and biological reality evolving around us is the subject of
this monograph.
Contents
1. Introduction
2. The Forces of Nature: A Universal Wave and Force Field
(UF) Related to the Chaplygin/Tangent Gas -- a Universal Source of Physical
Order
3. The
Emergence of Physical and Chemical Complexity:
A Critical Analysis of the Complexity Theory of Nicolis and Prigogine
4. A New Approach to the Emergence and Evolution
of Biological Complexity in the Aqueous Environment of the Living Cell.
4.1 Introduction
4.2 The Living Cell: An
Aqueous Medium
4.3
The Adiabatic Rupture of Cell Water and the Effect of this Entropy Change on
Biochemical and Genetic Reaction Rates
4.4 Chemical Reactions in the
Aqueous Environment of the Living Cell: An Entropy of Liquid Rupture
4.5
Entropy Increase, the 2nd Law of Thermodynamics and Evolution.
5. Conclusions on Biological Complexity: Do
Orderly Forces Acting on Random Kinetic Motions Result in the Emergence of
Variety on a Probabilistic Time Scale?
References

1. Introduction: Early theories of biological evolution, such as
those of Buffon, and Lamarck, stressed the obvious morphological and functional
adaptation that organisms have to their environment and asserted the
inheritability of acquired adaptations.
The theory was descriptive and speculative, but provided no physical mechanism
to bring about either the claimed adaptation or its inheritance.
Charles Darwin’s original
theory of evolution stressed the role of chance variations in automatically
fitting the organism to its environment, followed by the survival of the
fittest, and the passing on of this random variation in fitness to the
offspring. The theory was factually descriptive, and based on chance, but its
simplicity was a better fit to the canon of parsimony than earlier theories.
Mendel’s experimentally-based
genetics then emerged to assert the impossibility of inheriting anything other
than the genetic endowment. Darwinian chance variations were not inheritable no
matter how fit they were. With fixed genes, evolution could not take place.
Neo- Darwinism overcame the
Mendelian obstacle by postulating the random or chance emergence of new genetic
material, combined with the subsequent non-random probabilistic survival of the
genetically most fit population or sub-population. Given a sufficiently large
genome or pool of genetic potentialities, the probabilistic emergence of new
species would be automatically assured. The theory is quantitative via
probability theory once the necessary genome size is somehow generated. The
physical origin of the large genome pool is not rigorously addressed; instead
the automatic emergence of physical complexity is implied.
A serious attempt to explain
the role of entropy and the 2nd law of thermodynamics in biological
systems, where it appears to act in opposition to its role in non-living
systems, has been made by Brooks and Wiley [1]. These studies have not been widely
adopted, but they go directly to the heart of the problem in the search for a
physical theory of evolution.
Recent studies by the
Prigogine et al. school [2] have endeavored to properly ground the emergence of
physical complexity in physics and chemistry,
by studying the automatic emergence
of complexity from random physical and
kinetic motions when the random physical system is thermally stressed or not at
equilibrium. This approach properly addresses some of the physics, but fails
to include the role of the basic
physical forces involved – orderly,
non-random forces of gravitation fields,
orderly non-random inertial forces, orderly
pressure field forces, etc, etc, Thus,
its assertion that the desired
complexity simply emerges automatically from random kinetic systems is
invalid, since it leaves out the role of the
basic, non-random, physical
forces that also participate in bringing about
the new complex order on the observed probabilistic time scale.
In this present study, we
shall (1) examine the role of ordered physical forces arising from the
UF in bringing about physical
complexity, (2) examine the physics of the rupture of water -- the aqueous
environment of the living cell in which all biological reactions and processes
take place, (3) introduce a new entropy of rupture into cell reaction kinetics,
which in turn appears to be
reflected in the observed logarithmic time scale of evolutionary
history, (4) and, in addition to the
role of the 2nd law in evolution, we shall
introduce the possibility of two
additional entropy regimes or entropy orders arising in the Universal Field,
which appear related to the observed orderly
and complex characteristics of plant and
animal life.

2. The Forces of Nature : The
Universal wave and force field (UF) which is related to the Chaplygin/Tangent Gas: a
universal source of physical order, waves and forces
2.1.
The Forces of Nature
2.2. A
Universal Wave and Force Field (UF) Related to the Chaplygin gas and
Tangent Gas
2.3.
Wave Motion in the UF
2.4. The Universal Field (UF) and the Transmission of
Transverse Electromagnetic Waves in Space
2.5.
Maxwell’s Electromagnetic Waves and the UF
2.6.
Gravitational Force and the UF
2.7 The Strong
and Weak Nuclear Forces: Strong and Weak Shock Options.
2.8.
Summary
2.1 The Forces of
Nature: The four fundamental forces of nature are
gravitation, electromagnetic force, and the strong and weak nuclear forces.
Other subsidiary or related forces are the intermolecular chemical forces among
molecules such as the van der Waals and London forces, the force of surface
tension, pressure gradient force
etc.
The general definition of force is the space gradient of energy, or
F = dE/dx ( in
one dimension )
An
equivalent formulation is the dynamic or
Newtonian definition of force:
F =m (dv/dt) = ma
(in terms of particle mass m and acceleration a)
In
any compressible fluid the force is given by the Euler equation which, for
1-dimensional flow is
∂u/∂t + u ∂u/∂x + v ∂u/∂y
+w ∂u/∂z = −(1/ρ) ∂p/∂x
where p is the pressure, ρ is the specific
density and the term on the right hand side is called the
pressure gradient force.
Until
the emergence of the UF theory, the concept of force in physics has been
compartmentalized. Electromagnetism and the nuclear forces are conceived of as
involving some sort of “exchange of particles”. In the case of the
electromagnetic force, the particle exchanged is the photon, for nuclear forces
the particles are gluons or the Z particle, and so on. For gravity, a
“graviton” force particle has been postulated but has never been experimentally
detected. In short, attempts to unify the concept of force in physics have,
until recently, essentially failed.
The energy gradient
definition ( 1) requires a non-uniform region
in a field of energy where the force develops and acts on any material
particle at that point. The relevant
point we make here is that a field of energy and its space gradients is an
existing orderly physical element. The existing elements are not just the
molecular and atomic particles, but also include this force field which acts
jointly with them to produce a resulting complex motion. Complexity in such a
system thus arises automatically from the physics, but not solely from random
kinetic motions of particles; instead it arises from the interaction of the
random kinetic motions of the particles and the action of the co-existing
orderly force field.
The second or Newtonian
definition of force is phenomenological. It accurately describes what happens
but does not address the physical origin of the force. When the particle is not
accelerating there is no force present, when it does accelerate the force
emerges. Newton does not advance any hypothesis here to account for the full
nature and origin of the inertial force
that his equation so well describes, any more than he does with his
gravitational force F=m1m2
G/r2 which travels across the cosmos to tie all matter together in
orderly cosmic motions.
In addition to the four
fundamental forces there are other derivative, well understood forces of nature
such as the intermolecular forces which hold molecules together, the pressure
gradient force in gases (Euler Equation), the surface tension forces on
surfaces of liquids, vapour pressure forces. These are all ordered physical
realities. They are not probabilistic in the sense that they sometimes have one
value and sometimes another and that only their mean value is of physical
significance -- they are always orderly and determinate.
The attempt to unify the
various forces in terms of a grand unification theory has occupied physics for
generations. While its accomplishment eluded physicists, there was no
disagreement on the existence of forces as residing in quantitatively
understandable orderly physical fields.
Consequently, any attempt to understand the obvious emergence of
physical complexity should therefore take the orderly nature of forces into
account and not just focus on the random kinetic aspects of the material world.
The emergence of complexity or evolution is thus to be seen as a binary process
of probabilistic kinetics plus order.
It has proved possible to
unify the forces of nature by postulating a universal wave field (UF)
encompassing the Chaplygin/ tangent gas and its unique wave properties. This
universal wave field has emerged from cosmology, where the recent discovery of
the acceleration in the rate of expansion of the universe has quickly led to
the application of a compressible exotic or theoretical fluid called the
Chaplygin gas as a universal cosmic energy field [3,4,5,6,7,8,9,10,11].
Subsequent analysis of this
exotic cosmic fluid (especially in a version introduced by Tsien [4, 1939] and von Karman [5, 1941] called the ‘Tangent
gas’ in aeronautics ) plus the recent
formulation of an orthogonal, isothermal equation of state for the
field has led to the concept of a
Universal Wave field (UF) [ 12 -22} with
its remarkable wave and force
properties.
Since this UF entity is
compressible, it will yield a force identical to the Euler force equation
already mentioned.
2.2 A Universal Wave and Force
Field(UF) Related to the Chaplygin gas and Tangent Gas
The
UF has the following Equation of state relating the thermodynamic
variables p,v, ρ, and T
pρ = ART + B
; p = −Av +B
i.e. an adiabatic equation of state- corresponding to the Chaplygin/
tangent gas equation
pvk = pv-1 = const. with k = cp/cv = − 1;
n = 2/(k − 1)
and an isothermal equation of state ( which in the case of the UF is uniquely orthogonal to the adiabatic
equation)
p = +Av – B
The UF’s highly
unusual properties include (1)
the unique ability among known fluids or gases to propagate stable waves of
any strength, which also (2) uniquely
obey the simple classical wave
equation, (3) a unique ability to support transverse waves, which is
something impossible for any other known or theoretical fluid, and which thus
provides for the first time a physical basis for the existence of the
electromagnetic field and of
electromagnetic waves, and for
the transfer of radiation through space
at the speed of light, (4) the unique ability to carry purely attractive
gravitational waves which transfer gravitational force through space, and finally,
(5) a unique linear wave field
that may also serves as the basis for the transfer of quantum information
through space at quasi-instantaneous
speeds ( 1023 m/s). .
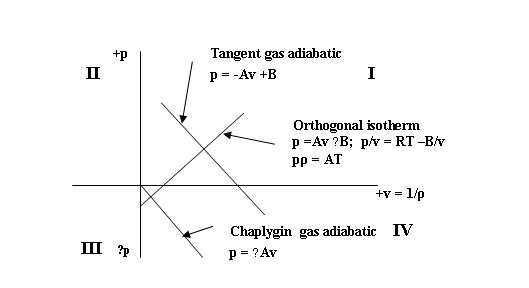
Fig. 1. Equations of State in the UF
2.3
Wave Motion in the UF
For simple
compressive waves the exact wave equation [8], expressed in
terms of the wave function ψ for amplitude, is
Ñ2 ψ =( 1/c2)
∂2ψ/∂t2 / [ 1 + Ñψ ](k
+ 1)
(1 )
or, for one
dimensional motion in the x-direction
∂2ψ //∂x2 = (1/c2) ∂2ψ /∂t2 / [ 1 + ∂
ψ /∂x ](k +1) (1a)
This exact
equation means that for material gases ( k > 0) compressive waves are always
unstable and grow with time. For very small amplitude waves, however, the term
in the denominator of Eqs. 1 and 1a involving ∂ ψ /∂x is approximately
unity, and the equation simplifies to becomes the classical wave equation [8]
Ñ2 ψ = (1/c2 )∂2ψ/∂t2
(2)
which has the general linear solution
ψ = ψ1(x – ct) + ψ2 (
x – ct)
(2a)
In the case of
the UF, however we see that, since k = –
1, the exponent ( k + 1) in the
denominator of Eqs. 1 and 1a becomes zero, thereby automatically reducing them to the simple classical wave
equation, but without the restriction to
small amplitudes necessary for molecular gases such as air.
The UF is
therefore truly unique in that it is automatically exact for
waves of any wave amplitude, large or small
and is not limited to infinitesimal waves as is the case with real gases. The UF is
therefore unique among gases since it is the only field in which the Classical
Wave Equation is strictly valid and which therefore can transmit stable waves,
of either condensation or rarefaction, of any amplitude.
So far we have not
distinguished between longitudinal and transverse wave motions in the UF.
Clearly there is no problem with stable longitudinal waves; they are uniquely supported in the UF,
and, moreover, they are not restricted to low amplitude acoustic type waves as
in real gases where k is positive.
We shall now present evidence
that transverse waves are
also uniquely supported in the UF and that they in fact correspond to
Maxwell’s electromagnetic waves.
2.4 The Universal Field (UF) and
the Transmission of Transverse
Electromagnetic Waves in Space
We should point out that this
is an entirely new concept which emerged only in 2005 after an analysis of the
isothermal properties of the exotic cosmic UF.
It has been known for over a century that real gases can only support
longitudinal waves, that is waves in which the density variations ±∆ρ
are along the direction of wave propagation. Real gases cannot support
transverse waves in which the density variations would be transverse to the
direction of wave propagation. It was this inability to transmit the transverse
waves of light which led to the demise of the old luminiferous ether
concept. We now ask: What is the evidence for transverse fluid
waves in
the Universal Wave Field (UF ) with its orthogonal adiabatics and
isotherms?
We consider a simple pressure
pulse ( ±∆p) in the UF as in Fig.6 below:
![]()
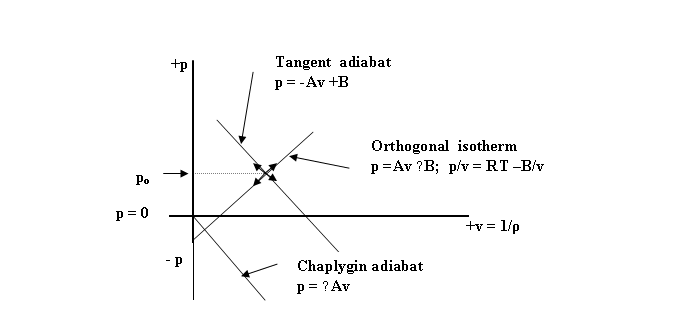
Figure 6. A pressure pulse ( ±∆p) in the
Orthogonal Environment of the UF
The initial or static state is
designated as po. When the pressure pulse ( +∆p) is
imposed from outside in some way, the UF must respond thermodynamically in two
completely orthogonal and hence two completely isolated ways, namely, by (1) an
adiabatic stable wave along the adiabatic ( TG) and (2) by an isothermal stable
pulse along the isotherm (OG).
Spatially, the pressure
disturbance ( +∆p) must propagate
in the direction of the initial impulse. But, since the two components of the
pulse are orthogonal, they must still remain completely independent and
physically isolated.
The only way possible for this to take place
is for the two mutually orthogonal components to also be transverse to
the direction of propagation of the two pressure pulses. Vectorially, this requires an axial wave
vector V in the direction of propagation
( say z) with the two pulses
orthogonally disposed in the x-y plane.
i.e. TG X OG = V, which is evocative of
the Poynting energy vector S = E x B in an electromagnetic wave.
E
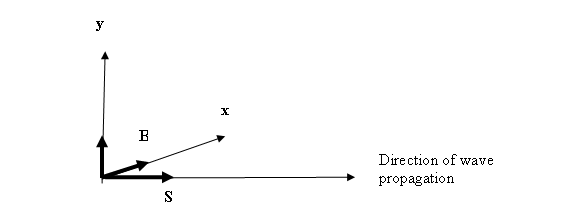
A wave of amplitude ψ
traveling in one direction (say along the axis x) is represented by the unidirectional wave equation
dψ/dx = 1/c dψ/dt
2.5
Maxwell’s Transverse Electromagnetic Waves and the U.F.
Here, however, in the case of
our adiabatic and isothermal pressure pulses,
we have two coupled yet isolated unidirectional waves, and this reminds
us of Maxwell’s coupled electromagnetic waves for E and B, as follows
dEy/dx
= (1/c) dB/dt and dBy/dx = (1/c) dH/dt
where c is the speed of
light, E is the Electric intensity and B is the coupled magnetic intensity.
Maxwell’s E and B vectors are also orthogonal to each
another and transverse to the direction of positive energy propagation.
Therefore, we have established in outline an
orthogonal two component wave system in
the Universal Field (k = −1) which formally corresponds to the E and B,
two component, orthogonal system of Maxwell for the propagation of electromagnetic waves through
space in a continuous medium. His equations for E and B are
Curl E = ∂Ey/∂x
= −(1/c) ∂B/∂t
(3)
Curl B = ∂By/∂x = − (1/c) ∂E/∂t (3a)
If we now designate our
Tangent gas as A ( for Adiabatic) and our Orthogonal gas as I ( for Isothermal)
then our analogous wave equations would be
Curl A = ∂Ay/∂x = − (1/c) ∂I/∂t (4)
Curl I = ∂Iy/∂x = − (1/c) ∂A/∂t
(4a)
The two systems are formally
identical. Therefore, we propose that the medium in which Maxwell’s transverse
electromagnetic waves travel through space
is physically identified with a Universal Compressible Field (UF) having the above described thermodynamic
properties for adiabatic and isothermal motions initiated in the UF by imposed
pressure pulses ( presumably by accelerated motions of electric charges.) The compressibility
property of the UF now properly accounts on physical grounds for the observed finite wave speed (speed of light c =
3x108 m/s)); moreover, wave motions in this fluid medium are
transverse as required by the electromagnetic observations.
It is possible to reduce
Maxwell’s two equations UF equations to a symmetrical single wave equation
∂2E/∂x2 = (1/c2) ∂2E/∂t2
(5)
∂2B/∂x2 = (1/c2) ∂2B/∂t2
(5a)
and similarly with A and I for our Adiabatic/Isothermal coupled wave in
the UF:
∂2A/∂x2 = (1/c2) ∂2A/∂t2
(6)
∂2I/∂x2 = (1/c2) ∂2I/∂t2
(6a)
This is not surprising since
the UF, with its k = −1 thermodynamic property, is the only compressible
fluid which automatically generates the classical wave equation ( Eqn. 2) with
its stable, plane waves. The formal agreement of the UF theory with Maxwell is
again striking.
Instead of taking our initial
external perturbation as a pressure
pulse ( +∆p) we should more realistically from the physical
standpoint take it to be a density condensation (s = ( ρ – ρo
) / ρo =
+∆ρ/ ρo). This will now result in a positive
pressure pulse (+∆p) appearing in
the adiabatic (TG) phase of the UF but a
negative pressure pulse ( −∆p)
in the isothermal or orthogonal perturbation component (OG) . This perturbation
is represented by the two orthogonal sets of arrows on the pv diagram, one
corresponding to +∆p and the other set corresponding to − ∆p.
As the wave progresses the two orthogonal vectors also rotate.
![]()
Figure 5. The physical
ambiguity which results from a pressure/density perturbation in the
Tangent/Orthogonal UF
An oscillating density
perturbation ( ±∆ρ) then results in an axial wave vector having two mutually orthogonal components of a density perturbation wave. This appears to correspond formally to the
Maxwell electromagnetic wave system with its two mutually orthogonal vectors
for electric field intensity E and
magnetic field intensity B.
We have thus established a case for the compressible
UF being a cosmic reality which generates and transmits transverse
electromagnetic waves through space.
These waves with their
oscillating space gradient of energy ( i.e. of force) generate the electromagnetic
force.
2.6
Gravitational Force and the UF
Here we shall not go into
details, but the analysis of the UF waves also provides for the first time a
system of exclusively attractive gravitational force as required by Newton’s
force equation.
Briefly, in any
compressible fluid, force is given by the Euler equation , which for 1-dimensional flow is
F = ∂u/∂t + u ∂u/∂x
+ v ∂u/∂y +w ∂u/∂z = −(1/ρ) ∂p/∂x
where the term on the right hand side is called the pressure
gradient force.
In any compressible
fluid medium, waves set up local transient
pressure forces. Most waves in real gases and fluids are pressure
oscillations (±∆p) and so they do
not exert any net directional force on a material object they encounter. However, in the UF special types of waves can
occur which can be either exclusively pressure compressions (+∆p) thereby
exerting a net repulsive force on any material body in their path, or they can be exclusively pressure
rarefactions (−∆p) which
would then exert exclusively attractive
force.
Consider Figure 6 where a condensation pressure pulse (+∆ρ; +∆ρ) imposed on the initial static pressure po
in the UF will produce a
rarefaction pressure pulse (−∆ρ) in
the isothermal mode of response. Consequently, a source of pressure
condensations will produce a train of isothermal pressure pulses as a response
in the UF, and these pulse will travel spherically outwards through space.
When these rarefaction waves eventually impact a material body
(mass) they will exert a net attractive
force on it. This mechanism therefore in simple outline is formally equivalent
to the force of gravity being produced by a rarefaction pressure gradient
force.
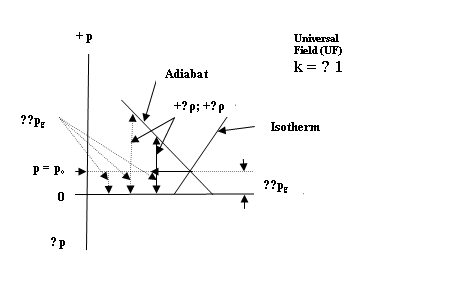
Figure 7. Positive density pulse ( +∆ρ) of any magnitude produces a quantized
gravitational rarefaction pulse (−∆pg ) of constant magnitude pg =6.67 x
10-11 kilopascals
2.7 Strong and Weak Nuclear Forces : Strong and Weak
Obligue Shock Options.
We are not
concerned with nuclear force in this monograph. However, for
completeness, we note here that they also are explainable theoretically in
compressible fluid flow theory via energy gradients which give rise to the
forces ( F = dE/dx ).
There is another interesting
compressible flow phenomena, that of shock discontinuities. In strong shocks,
the energy gradient in the shock discontinuity is very large, and so the
resulting associated force is also very large, and vice versa for the weak
shock phenomenon. Furthermore, in
oblique flow past an obstacle, the option may occur for either one or the other
shock to occur. This is depicted in the
so called “shock polar” which is a
representation of these two options in
a two-dimensional flow situation ( in
velocity coordinates u and v):
Weak intersection
W
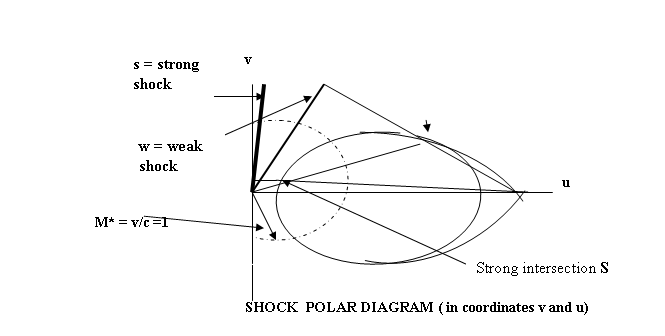
For each inlet Mach number M1 (
= VN1/c), and turning angle of the flow θ, there are two
physical options:
1) the strong shock ( intersection S) with
strong compression ratio and large flow
velocity reduction (p2 >>
p1; V2 << V1, and large spatial energy gradient and strong
force (Fs = dE/dx ) , or
2) the weak shock (intersection W, with small pressure
rise, small velocity reduction, small
spastial energy gradient and weak force
Which of the two options occurs depends on the
boundary conditions: low back, or downstream, pressure favours the weak shock
occurrence; high downstream pressure favours the strong shock.
It is natural to consider whether these two shock
options do not also correspond to the strong and weak nuclear forces. We point out, however, that the shock discontinuities in energy gradient ( dE/dx) which give rise to forces (F = dE/dx )
may actually have to reside in condensations of matter/energy and not in the UF itself. This is because in the UF the flow Mach
number M = v/c may approach, but never reach or exceed unity, and
therefore shock discontinuities or instabilities cannot occur
in it; the UF supports only stable wave forms,
although they may be of any strength, strong or weak The details of the nuclear forces do not
concern us here.
2.8. Summary
Other
minor forces exist, such as those of surface tension, intermolecular force and
so on, but they are all variants of the fundamental forces set up by spatial
energy gradients or by accelerations in the UF.
It
may be worth restating here that this remarkable ability of the UF to unify all
the forces of nature arises from its property that changes the sign of k in the
equation of state pv k = const. from k = +1 ( ideal isothermal
gas) to k = – 1 ( UF ). That gives us the UF orthogonal and tangent
gas equations of state p = +A
+B and p = −A +B, while the
wave equation simply becomes the linear classical wave equation ∆2
ψ = ( 1/c2 ) d2ψ/dt2. from which
the forces emerge. All thus unification
and simplification arising from a simple change of sign in the ratio of the
specific heats k from positive to
negative is truly astonishing.
This then completes our
survey of the orderly wave and force field.
We now return to the emergence of complexity in physical systems and
then to applications to biological evolution.

3. The Emergence of Physical and Chemical Complexity.
A critical analysis of the complexity examples of Nicolis and Prigogine
In their book Exploring Complexity, Nicolis and Prigogine [2] examine in detail the
emergence of physical and chemical complexity from previously random kinetic systems such as gases and
liquids when some source of additional energy is applied to the system in a
non-equilibrium process which they
term stressing
of the system.
Various physical forces that
are involved are mentioned, intermolecular forces and gravitation in
particular, but they are treated by Nicolis and Prigogine simply as “givens” or
adjuncts to the random kinetic motions of the atoms and molecules involved.
This limited approach to the nature of the forces that cause the ordered
motions and interactions leads to overlooking the orderly nature of the ever-
present force fields that exist in each of the examples of complexity they
present, and so to their faulty conclusion that biological complexity arises
solely from random or kinetic physical events.
3.1.
Their first physical example is the ‘ production of an ice crystal’ ( e.g. a dendritic snowflake ) from bulk water. They
state that when the water is cooled below the freezing point the snowflake
appears and grows. This, they argue is a
simple example of the emergence of a highly ordered state from a disordered
state, i. e. complexity emerges automatically upon cooling a liquid below its
phase change temperature
Comment: Their first example is presented simply and makes no
claim to exactness. However, we should point out that the process of snow flake
production from water involves, not bulk water, but disordered water
vapour. At most temperatures, the
formation of crystalline ice from either
bulk water or water vapour requires the presence of a suitable substrate or foreign ice nucleus
whose preexisting orderly structure is essential to the initiation of the ice
formation. Once nucleated, the type of crystal produced from water vapour is,
moreover, not typically a dendrite, but depends instead upon the ambient
temperature and pressure of the water vapour field surrounding the growing
crystal. The crystal forms may range from needles to plates, to columns, to columns
with plates attached to their ends, to
dendrites and to other composite shapes
depending on the ambient temperature and vapour pressure [24,25,26,27 ]. In addition, heat of sublimation is released
in the change of state from vapour to solid ice and this evolved heat must be
taken up by the total system as an entropy increase ( i.e a disorder
increase). The vapour flows are governed
by the vapour pressure gradient forces arising from the gas density gradients at
the points where the water vapour interacts with the emerging crystal shape.
The forces are not random and kinetic, but are orderly and determined by the
gas law. Thus the resulting ice crystal shapes, once initiated by the
pre-structured ice nuclei, are jointly determined by random or kinetic
behaviour, by the orderly pressure gradient forces of an ideal gas, by the
ambient temperature, and by the crystal energy requirements of the solid state
that emerges. (Homogeneous ice
nucleation can take place at around -40C but the argument is not essentially
altered).
Clearly,
the complexity which emerges upon ice crystal formation does not arise
automatically from a purely kinetic state, but instead involves both a random
kinetic component in the vapour and an orderly field of short range vapour pressure
gradient forces around the growing ice crystal.
(The force of gravity in this case acts only indirectly. But it should
be remembered that it does produce the orderly ambient pressure field of the
atmosphere, including its partial water vapour pressure component at the
particular height above ground involved) .
Hence, in this case,
complexity emerges, not automatically from kinetic disorder by
self-organization as Nicolis and Prigogine concluded, but instead from an interaction
of disorder and order. The orderly gravitational force produce an ideal gas
field; the orderly vapor pressure gradients surrounding the random nucleating event produces an
orderly snow crystal; the orderly crystal has a random element or sub-structure which manifests itself in a
great variety of individual variations
within the general crystal type dictated
by the prevailing temperature and humidity of the vaporous environment ( 24,25
].
3.2.’ Convection patterns
in thermally disturbed fluids: Bénard cells’
This
case is called by Nicolis and Prigogine
“a prototype of
self-organizational phenomena in
physics” and so it will bear our careful analysis.
They
discuss a shallow layer of water between two extended top and bottom plates.
Left to itself the water rapidly assumes an equilibrium state of uniform
temperature or uniform internal kinetic molecular disorder. If the system is
now heated from below by a small amount, the injected heat is transferred
upward by thermal diffusion, that is by the kinetic molecular motions under the
action of the physics of a diffusion wave.
Next, when more strongly heated by a certain critical amount, the
temperature stress ∆T from bottom to top will set up a regular pattern of
convection (Bénard) cells consisting of rising and descending fluid streams
which organize into rotating cells. The direction of rotation seems to be
random. This complexity or pattern has thus emerged automatically when the
uniform state is stressed thermally and an energy flow is set up. The rising
and falling motions are the result of density (buoyancy) differences in the
various parcels of water, i.e. from assemblages of molecules large compared
with the size of the individual molecules which are still undergoing only
random motions. The fluid motions arise from the instability of dense and less
dense water parcels side by side at a given level resulting in a descent of the
more dense and the rise of the hotter ( less dense) parcels.
Various
patterns of convection cells result depending on the boundary conditions and
the vertical gradient of teméperature stress. For a sufficiently large heating,
turbulent or disordered motions on the macroscopic scale set in.
This
type of convective motion is treated in detail in texts on dynamical meteorology
under the topics of potential instability and
cumulus dynamics, where the
density differences of perturbed air parcels under a uniform gravitational
force field, combined with differential
wind shear variation with height, explain the resulting cloud patterns and
motions.
Nicolis and Prigogine conclude from their analysis
that:
“ To summarize, we have seen that
non-equilibrium ( heating) has enabled the system to avoid the thermal disorder
and to transform part of the energy
communicated from the environment into an ordered behavior. We can
therefore cay that we have witnessed the birth of complexity. Our complexity
achieved is rather modest, nevertheless, it presents characteristics that
usually have been ascribed exclusively to biological systems. More important, far from challenging the laws
of physics, complexity appears as an inevitable consequence of these laws when
suitable conditions are fulfilled.”
Since
the direction of rotation in the cells can be right handed or left handed and is
apparently random, they also introduce the notion of chance at this
point and draw a comparison between this
and the randomness or chance asserted to exist in biological evolution where
mutation is asserted by them to be controlled by chance and natural selection
operates. No physical mechanism for this connection between physics
and biology is offered.
On
thermal convection phenomena they postulate (1) an initial uniform ,
kinetically disordered layer of fluid,
plus (2) a thermal stress imposed on a
boundary which sets up a gravitationally unstable stratification, and
then at some critical value this results in ordered convection cells
forming; this they interpret as being
the automatic emergence of physical complexity from disorder.
Comment: We have several physical elements here: (1) a compressible fluid initially uniform and
then (2) stressed or disturbed thermally to bring about an unstable
stratification when (3) a suitable gravitational field also
exists. Nicolis and Prigogine miss the
importance of the presence and strength of the gravitational field. It is crucial to the emergence of the
particular convection they cite. For example, if the gravitational field is
absent, say in a space ship in outer space, no potential motion can exist and
no cells can form. Again if the field is too weak, the cells can never reach
the critical stress value. Again if the field is not uniformly vertical , but
is converging or diverging, an entirely
different complexity pattern and motion emerges. This specifically orderly
gravitational field element is central to the existence and nature of the
convection cell phenomenon, The phenomenon is therefore the physical result of
the interplay of random kinetic molecular motions, ordered by gravity into an
orderly pressure field (pressure of the fluid) plus the introduction of an
asymmetrical temperature pulse, plus the action of an orderly gravitational
buoyancy force field. The resulting complex behaviour then emerges, not from
chance as they assert , but from the interplay of random or chance, probability
laws, induced asymmetry and an ordered force field.
Thus,
the Benard and convection cell
phenomenon in their example is not one of emergence of complexity and order
from physical randomness alone; instead,
it is a complex physical interaction of factors, a stressed physical disorder
plus an orderly physical force field. Physical complexity does not arise solely
from disorder but from an interaction of
disorder and imposed order. The Nicolis
and Prigogine “prototype of self organization phenomena in physics” example is fundamentally flawed and
incomplete as a foundation for their
emergence of physical complexity thesis.
(Properly
formulated, however, the Bénard cell
example does show central
features of the emergence of physical
complexity in the physical world. Thus, they have chosen the proper
example, even although it does not support their conclusions) .
3. ‘Self Organization
Phenomena in Chemistry: The
Belousov-Zhabotinski (BZ) Reaction’
Nicolis
and Prigogine consider the state of
equilibrium of any chemical reaction
reached when two reactants
produce a third chemical substance, or
more typically two more substances
A + B → C
+ D and the reverse reaction
C +
D → A + B
They
then point out that, when certain of these reactions are disturbed or
suddenly moved far from equilibrium,
quite unexpected results are obtained
They
then cite the BZ ( Belousov- Zhabotinski) reaction. For example, a solution of cerium
sulfite Ce2SO4, malonic acid CH2(COOH)2
and potassium bromate KBrO3 , in sulfuric acid, with an excess of Ce4+ ions gives a
pale yellow colour, while an excess of Ce3+m ions gives no
colour at all.
If
now the reaction is carried out with stirring, several quite different types of
behaviour are observed, namely oscillatory or clock-like behaviour of alternate
colour and absence of colour, and chaos or turbulence.
If
the reaction is carried out without stirring, orderly propagating wave fronts
bring about the formation of spatial coloured spirals and spatial target
patterns.
They
then cite this behaviour as exemplifying the emergence of complexity from
purely random or chance motions.
Comment
In
their analysis of the B-Z chemical reaction
Nicolis and Prigogine fail to discuss the actual forces
involved. In chemical combination
processes such as chemical reactions the
relevant forces are intermolecular.
These are Van der Waals forces arising from chemical dipoles existing in
the asymmetrical atoms and
molecules. It is these intermolecular
forces which act to hold the various reacting
chemical species or molecules
together to form new chemical reaction products once they succeed in
colliding with sufficient energy and
entropy of activation. Without these
forces of attraction, the molecules would simply collide and rebound
kinetically and randomly as in a gas or
liquid without combining at all.
However
we have shown that all forces are orderly and arise from ordered deterministic
force (linear and non-linear) and
so their thesis that complexity is the
automatic emergence of order, and pattern
from random or chance subsystems alone
is incorrect.
[Note.
Later in Section 4.3 we discuss the
entropy change in liquid ruptures that
occur during reactions in solution and which are intimately connected
with chemical reaction kinetics. They arise from ordered forces in a
compressible medium, and have a key role
in determining the rates of the chemical reactions; they are not
described here as they are not essential
to the present example].
3.4.’ Surface Tension
Induced Phenomena in Materials Science’
Here
they mention interfacial phenomena such as droplet formation and interfacial flows as examples of complexity emerging
spontaneously from physical uniformity, but give no analysis and do not discuss
the force of surface tension which again is ordered and not random..
3,5. ‘Cooperative
phenomena induced by electromagnetic fields: electrical circuits, lasers,
optical bi-stability’
The
example offered here is the action of a coherent electromagnetic field ( a
laser beam) injected into a resonant cavity
filled with a suitable light absorbing medium. Under certain conditions
the behaviour becomes bistable and can act as on optical switch. The behaviour
is complex and non linear.
Comment: While the behaviour is complex,
it is not the result of any spontaneous emergence of complexity from a
non-complex or random predecessor state. Rather it is the result of a random
(uniform) medium being acted upon by a highly-ordered electromagnetic
field force.
The
motions and forces in the e/m field are highly ordered. They are given by the
Maxwell electromagnetic wave equations
Curl E = ∂Ey/∂x = −(1/c)
∂B/∂t
Curl B = ∂By/∂x = − (1/c)
∂E/∂t
Consequently,
complexity arising in these systems must also reflect these highly ordered
waves and their forces. It can still properly
be called self-organization, because it is an entirely physical process, but it is not a chance
mechanism. As we have seen in Section 2, the electromagnetic force arises from
the occurrence of transverse waves in the UF, the fundamental field of physical order.
3.6. ‘Complexity in Biological Systems’
Here
they state, “ Being convinced by now that ordinary physico-chemical systems can
show complex behaviour presenting many of the characteristics usually ascribed
to life …” They then describe in very general terms a few examples of
biological systems.
This
appears to be a greatly simplified claim. It might be more accurate to say
“presenting some of the most elementary complex characteristics of living
systems.”
Of
course since biological systems undergo the same physical and chemical
processes as do non-living systems, it is only natural to suspect that they
also exhibit the same rudimentary
complex characteristics that arise in non-living systems. The problem is not
that biological systems are not as
physical as non-living
systems, but that they are so outwardly different
from physical systems, and that we cannot presently explain why this is so in the necessary detail. In particular, so far as entropy flow is
concerned, biological systems appear to violate the 2nd law.
3.7. ‘Complexity at the Planetary and Cosmic Scale’
This
is an interesting listing of complexity of physical events but the discussion is never at the
level of the physical processes that occur. While it is open to an analysis
that would include the details of the
forces involved, it is not essential for our purposes and is set aside for some possible future
exploration.
3.8. ‘The mathematical theory of complexity’. The bulk of the Nicolis and Prigogine analysis goes
on to deal in some depth, with the mathematical theory of complexity, chaos and
randomness. This will be very useful once the necessary clarification of the
role of orderly forces that we are recommending is completed, so as to provide
a formulation of a more complete theory of complexity, both physical and biological.
3.9 Conclusions on Physical
Complexity
The
general process under study is the action of fields of force on material
particles (atoms and molecules) and on material assemblages of matter (gases,
liquids and solids). In the absence of external force fields, the particles
undergo random or purely kinetic motions.
When orderly field forces are present they interact with the kinetic
field and a composite pattern of order or complexity then emerges from the random
chaos. It is our thesis that this binary, composite character of complexity
must be considered in any account of the evolution of the physical universe and
the biological world.
It
should perhaps be noted in addition, that, with all physical force apparently
arising from as universal field UF, this same field is uniquely orderly: (1) It is the only wave field that obeys the linear classical
wave equation; (2) it alone supports and
transfers stable waves of any amplitude; (3) it alone supports waves of both
compression and rarefaction; (4) it
alone accounts for the forces of nature and uniquely unifies them; (5) it alone
supports transverse electromagnetic waves in space. Those unfamiliar with gas dynamics, acoustics
and compressible flow may not fully realize how astonishing this all really is,
but in fact, apart from the UF ( i.e. the Chaplygin/Tangent gas) in the
physical world no kinetic molecular gas or compressible fluid can support stable linear waves of any finite amplitude
whatever. All gases or compressible fields other than the UF are non-linear,
unstable and tend towards shock discontinuities. If then, a unique orderly, stable, linear UF
wave and force field underlies all the molecular physical world, and is the
source of all its forces, then the
proposition that complexity arises automatically and solely from
molecular disorder and stress energy is untenable.
Many
other physical interactions of force and kinetic interaction are of interest
and remain to be studied. In the
meantime we turn to biological complexity.

4. A
New Approach to the Emergence and Evolution of Biological Complexity in the
Aqueous Environment of the Living
Cell Involving an Entropy Rate Factor
7N
4.1 Introduction
Up to this point, we have argued
that complexity in the physical world results from an interplay of two elements, namely, random or kinetic elements
on the one hand, and orderly forces on the other. Complexity is thus a binary
physical process. In the following approach to explaining complexity in the
biological world of living organisms, we shall cite these same two physical elements and their
interaction as agents in bringing about the observed complexity.
We examine biological processes
and evolution within the basic living cell. We concentrate on the physical forces that must be present to operate
the metabolism of the living cell, to operate on the proteins and genes in the
body cells so as to enable cell growth, the transmission of the organizing
instructions to the cell component parts, and to operate on and occasionally to
mutate or change the genes so as to transmit the biological integral forms from
one generation to the next while still allowing for emergent change or evolution..
Clearly this approach is
different from that of neo-Darwinian population genetics, which offers little
on the physical processes underlying the emergence of the gene forms themselves
and focuses mainly on the results of various probabilistic combinations within
the given, already existing, very large gene pool. Physical process involved in
conformational protein alteration and gene mutation, while involving
probabilistic process, cannot be essentially or exclusively random since they
involve orderly forces.
Our new physical approach also involves a closer look at the
aqueous environment of the living cell. We look at biological interactions and
reactions as taking place in an
aqueous medium, that is, as chemical reactions occurring in suspension or in
solution in the cell aqueous medium.
We shall first look at a
neglected necessary physical step in any reaction in water, namely the local rupture and
removal of the water films between reactant molecules in the living cell so that any collision between molecules can
come to completion and allow the reaction to proceed. This new element of cell
water rupture is then included in the energy of activation in standard
collision theory of chemical reaction kinetics, or in the entropy of activation
in the statistical mechanics approach to chemical reaction kinetics.[28].
4.2. The Living Cell: An Aqueous Medium
The
living cell is typically quasi-spherical in shape and from 10 to 30 micrometers
( 10-5 to 3 x 10-5
m) in diameter. The cell is surrounded by a cell wall or semi-permeable
membrane of lipids about 90 angstroms thick, which serves to isolate the cell
from its environment so as to maintain its identity or homoeostatic nature. The
cell membrane is reminiscent of the physical prototype of a lipid envelope in
water- oil emulsions, or of the monomolecular films of various terpeneoid and
essential oils from vegetation that coat atmospheric cloud droplets in most
areas of the world.
The
aqueous cell medium. The typical living cell is essentially a water
globule, surrounded by a semi-permeable cell wall or membrane, and having in
solution or suspension various organic structures, and with a central core of
genetic material which directs the cell life, growth and reproduction. The essentials of life therefore take place
in water, so that we must consider the physical properties of water in any
biological process or function; it is not simply an inert physical medium.
Furthermore, at
the basic physical level, any reaction or interaction in a liquid must involve the usual elements of (1) collision energy of activation ∆E and
(2) the entropy of activation ∆S , both of which quantities are standard
in chemical reaction theory [28]. It is
suggested here that this entropy of activation also must include the energy
involved in the rupture of the water film around or between the various
suspended or dissolved chemical reaction species. That is to say, any liquid
film that separates two reactant molecules must first be ruptured and removed
before actual collision and subsequent reaction can occur. Only after this
rupture has taken place and a ‘void’ has opened up can the chemical
/molecular constituents collide,
interact or reassemble.
The cell must obey the thermodynamics of the physical fields it
encloses, in particular this means for biology that the entropy changes in the
cell and in particular the direction of such changes must be considered. These
may conveniently be summarized on the usual pressure volume diagram of
thermodynamics as follows:
(1). Micell or Lipid-covered Water Droplet ( A Physical Analogue of the
Living Cell
Properties

Simple ‘cell’ wall
Aqueous with random mineral species
in solution
No fixed size. Size is
determined by relative humidity of ambient vapour and surface tension
Minimum interior complexity
Non-living, non-reproducing,
i.e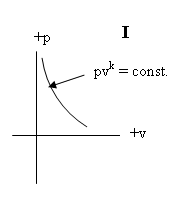 . a simple kinetic physical system
. a simple kinetic physical system
Equation of state has form pvk = const. (Quadrant I )
Internal de Broglie waves in
aqueous fluid
Pressure, specific volume,
density, temperature and pressure energy are all positive
Entropy change dS
= dQ/T is always positive and the 2nd Law of Thermodynamics holds
This order of entropy change( i.e. the 2nd
Law of thermodynamics) governs gas dynamics as it impacts on cosmic
evolution
(2)
Animal (plant) Cell
 Properties
Properties
Complex cell
wall
Aqueous with complex inclusions
(Nucleus, orgnelles,
DNA/RNA etc)
Cell size 20-30 micrometers
Great interior complexity
Living
and reproducing
Supports enclosed UFI standing wave
forms in Tangent gas and Orthogonal (isothermal) gas
Eqn. of state: Tangent gas
p = −Av +B (Quadrant I)
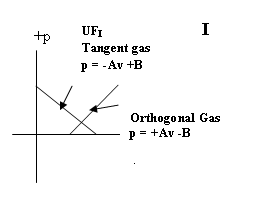 Eqn. of
state: Isothermal (Orthogonal) gas p =
+Av −B
Eqn. of
state: Isothermal (Orthogonal) gas p =
+Av −B
Entropy change in the
Tangent gas, ds = dQ/T is positive, and
2nd Law holds( i.e. “uniform disorder seeking”)
Entropy change in the Isothermal
(Orthogonal) gas is zero, ds = 0/T = 0,
( i.e. “ stability seeking” )
Pressure positive (+p)
Specific volume positive (+v)
Specific density
positive (+ρ) ( i.e.Wave forms are compressive)
Pressure energy is positive (
+pv)
It appears that the entropy
change laws in Quadrant I of the p-v thermodynamic diagram would then enter
into the nature and direction of plant and
animal evolution as follows:
Quadrant
I:
(a) Tangent Gas: dS = dQ/T is
positive ( 2nd law of thermodynamics holds ( “disorder seeking”)
(b) Orthogonal ( Isothermal) Gas: dS = dQ/T = 0 ( “stability seeking” )
Note: There is another entropy condition in the UF , namely that
in Quadrant II dS = − dQ/T, which
has very unusual properties apparently matching the human condition. This is discussed briefly in Section 5 below,
and in detail in Science and the Soul/Body Problem:
An Exploratory Reassessment.
4.3 The Adiabatic Rupture of
Cell Water and the Effect of this Entropy Change on the Rate of Biochemical and Genetic
Reactions
The process of
the formation of microscopic voids or micro- bubbles in a fluid which may lead to
its rupture has been intensively studied in the phenomenon of cavitation and
bubble formation, and in the reverse process of the homogeneous and
heterogeneous nucleation of condensation of
liquid from the vapour [28].
However, with
respect to liquid rupture, we must take note of the fact that there is a
long-standing major discrepancy between the theoretical and the experimentally
observed tensile strengths for all liquids, and especially for the case of
water. Recent work has offered a possible solution to this long standing
problem by postulating an adiabatic
rupture process instead of the usual isothermal one; this new approach has
succeeded in a reconciliation of theory
with observation. The new theory is as follows:
---------------------------------------------------------------------------------------------------------------------------------------------------------------
Adiabatic
rupture as an explanation for the anomalous weak tensile strengths of liquids
and solids
Bernard A. Power
Reviewed Aug. _ Sept. 2007: Revised Oct. 2007
The observed tensile strengths of liquids and solids are orders of magnitude lower than the theoretical isothermal rupture values. The discrepancy is currently explained by heterogeneous nucleation of the ruptures in the theory of nucleation rates. Still, the observations for water do not agree with current theory. However, an adiabatic rupture producing of voids or bubbles ( Equation of state pvk = const.) would give much lower theoretical tensile strengths in agreement with the observations.. The concept should be of interest to materials science, to chemical reaction kinetics in aqueous solution, and so to cell biology and genetics.
________________________________________________________________________
1. Introduction
Theoretical estimates of the tensile strength of solids and liquids give values of around 3 x 104 to 3 x 105 atm.. However, for solids, the experimental values are around 100 times smaller than that, while for liquids, the observed values are 600 to 1500 times smaller at 50 to 200 atmospheres (Kittell, 1968; Brennan ,1995), with water being among the very lowest.
A simple classical derivation (Frenkel, 1955; Brennan 1995) of the theoretical tensile strengths of solids or liquids considers the fractional volumetric expansion ratio ∆V/Vo needed to form the rupturing void, and this then is equated to an average numerical value of about 1/3 . Then, since liquids and solids have compressibility moduli K which are about. 105 to 106 atmospheres, we have a rupture pressure p(max) = −K(∆V/Vo). Taking the average 1/3 value for ∆V/Vo , the rupture pressure p(max) then becomes the theoretical 3 x 104 and 3 x 105 atmospheres just mentioned, far higher than actually observed.
For solids, the discrepancy in tensile strength is usually ascribed to heterogeneous nucleation of rupture at defects such as cracks or dislocations in the lattice (Kittell, 1968). In the case of liquids, the even larger discrepancy is usually explained by invoking the presence of irremovable tiny gas or solid nuclei within the liquid, which act to lower the pressures and tensions needed for mechanical rupture. Still, there remain discrepancies, and the foreign nuclei explanation, or heterogeneous nucleation process acting alone, has appeared somewhat artificial, especially since the thermal rupture ( boiling) values do agree more with the theory.
2. Adiabatic
cavitation
The basic mechanical equilibrium equation for the production of a spherical void, or vapour-filled bubble, in a liquid by rupture is usually expressed as a balance of forces inside and outside the spherical incipient void :
![]()
![]()
![]() pB
− pL = ∆pmax = 2 σ /RC (1)
pB
− pL = ∆pmax = 2 σ /RC (1)
which gives the relationship between the (negative) rupture pressure ∆p(max), the interfacial surface tension σ, and the rupture radius r. This process is also assumed to take place at the temperature of the bulk liquid, that is to say isothermally.
The formation of a bubble by rupture thus requires a negative pressure ∆p(max) exceeding the tensile strength 2 σ/r in order to create the spherical void. However, instead of the isothermal process ( with general form of its equation of state pv+1 = const.) which gives those unobserved high tensile strengths and rupture predictions, we could conceivably have an adiabatic rupture with pvk = const. A, where k > 1. With k greater than unity, the adiabatic rupture pressure ∆p(max., adiabatic) will always be less than the presently assumed isothermal rupture pressure.
To see this more clearly consider the following:
The isothernmal bulk modulus or modulus of elasticity for a liquid K is given by
Kis = − v ∂p/∂v
And the adiabatic modulus is
Kad = − v ∂p/∂v= k p where k is the adiabatic exponent or ratio of specific heats cp /cv
For liquids ( e.g. water )the two moduli have nearly the same numerical value.
The pressure at the critical point is then
p(max.) = −Kis (∆V/Vo) and
p(mac.) = − Kad. (∆V/Vo)
The adiabatic bulk modulus Kad. for water has the value 2.2 x 104 atms. Table 1 then shows the effect of taking the adiabatic rupture/cavitation mechanism in water over a range of values of (∆V/Vo) i.e. (ρ/ ∆ ρ ) and for various values of the adiabatic exponent k from >1 to 7.. We point out first that V is the reciprocal of the density ρ, and so we can put . (∆V/Vo)k = (ρ/ ∆ ρ)k which is more convenient., that is
P(max.) = − Kad. (∆V/Vo)k = − Kad(ρ/ ∆ ρ)k
(
The first step is the conversion of the liquid water in a small volume V to a “gas-like” structure at the critical point, which means a fractional volume expansion of about 0.333 (i.e. the density of water at the critical point drops from 1 to about 0.3333). This initial step obviously requires the injection of a sufficient energy. The rupture pressure in the new gas-like volume at this critical stage is now p(mac.) = − Kad. (∆V/Vo) = 2.2 x 104 (0.333) = 7326 atm.
The second step is the adiabatic expansion of the same ‘gas-like’ volume to a larger bubble volume with consequent decrease of the pressure. Clearly, for any given expansion ratio, the adiabatic expansion yields a much smaller final rupture pressure than the usual isothermal rupture model. For example, in Table 1, a volume expansion of 1/3 (density ratio ρ/ ∆ ρ of 0.333) yields an isothermal rupture pressure of 7326 atmospheres, while the adiabatic expansion at k = 7 has a rupture pressure of only 10.1 atmospheres .(The experimental data also show a definite effect of temperature on the final rupture pressure; this does not affect the conclusions reached here, since they are based on comparative values of the isothermal and adiabatic processes at any given initial temperature).
Table 1
Adiabatic rupture pressure p ( max.) for water ( Kad. = 2.2x104 ) for various assumed values of density change ratio (ρ/ ∆ ρ)
Rupture pressure (p ( max.) ( p = KAd. (ρ/ ∆ ρ)k)
(Atmospheres).
Density
Ratio
(ρ/ ∆ ρ)
k = 1** k = 2 k = 3 k = 4 k = 5 k = 6 k = 7
0.1 3300 atms. 220 22 2.2 0.22 0.022 2.2x10-3
0.20 4400 880 176 35.2 7.04 1.41 0.28
0.30 6600 1980 594 178 53.5 16.0 4.81
0.3333* 7326 2444 815 272 90.5 30.2 10.1
0.40 8800 3520
1408 563
225 90.1 36
0.5 11000 5500
2750 1375
688 344 172
0.6 13200 7920
4752 2851
1711 1026 616
1 2.2x104 2.2x104 2.2x104 2.2x104
2.2x104 2.2x104 2.2x104
*. Density ratio (ρ/ ∆ ρ) at the critical temperature TC for water is approximately this value of 0.33, the same value assumed by Frenkel
** Quasi-isothermal
Clearly, the isothermal hypothesis fails to yield the observed rupture pressures of around 50 -250 atmospheres for water at any assumed density ratio. The adiabatic expansion hypothesis, however, does let the pressure reach the experimentally observed low values.
What value for k are we then to adopt for pure water ? At the critical density expansion ratio of 0.333, any value of k from k = 4 to k = 6 would encompass the observed ed rupture pressures of about 250 to 50 atms. However, it may also be valuable to revisit the value of k = 7 obtained by Courant and Friedrichs (1948) who discussed the expansion and contraction of spherical blast waves in water, and fitted the experimental data to a quasi-equation of state for water under a pressure of around 3000 atm., which is pv7 = const or p =A ρ7 + B. They also derived this same value of the adiabatic exponent k = 7 theoretically as a solution to their non-linear flow equations for purely spherical ( i.e. radial) shock expansions in fluids. Their evidence that water rupture, at least in explosions, is spherical and adiabatic would also seem to be generally applicable, since all ruptures, even non- explosive ruptures, are quasi-sudden, and so, at least initially, they all could be adiabatic as well.
As to the proper value of the density ratio (ρ/ ∆ ρ) to accept, if the rupture process for water were envisaged as taking place by a transformation from its usual density of 1 by one of the usual cavitation mechanisms, such as a burst of electromagnetic or acoustic radiation into a small liquid volume ( the radiation being energetic enough to break all the liquid water bonds in that volume quasi-simultaneously), we would have a “ gas-like” liquid suddenly emerging with an expansion ratio of 0.333. Once the ‘gas-like volume has emerged, we see that it must at once expand from an initial gas-like density ρ, again taken as unity, to some smaller gas-like density ∆ ρ. by either the isothermal route p = K ((ρ/ ∆ ρ) or the adiabatic route p = K (ρ/ ∆ ρ)k where k is now greater than unity. The density ratio must then fall from unity to some value consistent with the usual equation for pressure equilibrium, p(max) = 2σ/r., where r is the radius of the critical bubble size.
Clearly the isothermal hypothesis cannot reach the observed low rupture pressures of 250 atmospheres or less,, while the adiabatic process can. From Table 1 we again see that a k value of 7, over the range of density expansion ratios (ρ/ ∆ ρ)k .from 0.4 to 0.6, would more than encompass the observed range of rupture tensions of 50 to about 250 atmospheres at normal temperatures.
The proposed model would l require simultaneous radial rupture over a sufficient number of adjacent bonds, and therefore the theory of nucleation rate analysis would still appear to apply. The radial rupture might also of course be heterogeneous, and then all the various heterogeneous mechanisms of bubble formation presently considered may still be in play.
The proper value to be used for k in aqueous solutions, where the densities are different from those of pure water, would appear to be a matter for further study.
The third step: the
attainment of a critical radius rc for rupture
I must be noted that Step 2 above is based solely on the density ratio ρ/ ∆ ρ and has not specified any actual initial or final density or ( specific volume. ) However as the “gas-like’ liquid bubble expands, it eventually must physically become an ordinary vapour –filled bubble of homogeneous nucleaton theory, and the latter theory requires that, for the bubble to persist, it must meet the critical stability condition:
pB − pL = ∆pmax = 2 σ /RC
Table 2 shows this final stability condition over a range of sizes , rc
Table 2
Critical ( stable) radius rc for various rupture pressures
in water
Critical radius of bubble, rc Rupture pressure, p(max) = 2 σ /r
(cm) (m) (σ = 75 dynes/cm)
a) (dynes/cm2) b) atmospheres (dynes/cm2 x 10-6 )
1 cm 0.01 m 140 1.4 x10-4
10-1 0.001 1.4 x 103 1.4 x 10-3
10-2 10-4 1.4 x 104 1.4 x 10-2
10-3 10-5 1.4 x 105 1.4 x 10-1
10-4 10-6 1.4 x 106 1.4
10-5 10-7 1.4 x 107 14
10-6 10-8 1.4 x 108 140
10-7 10-9 1.4 x 109 1400
10-8 10-10 1.4 x 1010 14,000
Notes:
1. The ratio between the critical state liquid pressure ( 1.4 x 104 atms).and the observed average rupture pressure for water ( say 150 atms) is about 100/1.
2. On the isothermal expansion hypothesis with p1/p2 = V2/V1 , the volume ratio at critical rupture must be the same i.e. about 100, .so that the radius ratio is r2/r1 = 1001/3 = 4.64.
On the adiabatic expansion hypothesis ( with k =7), it becomes p1/p2 = (V2/V1 )7 , so thatV2/V1 = (p1/p2)1/7 = 1.93. and r2/r1 = (1.93)1/3 = 1.25
3. If a bubble is to reach the critical rupture size of 10-8m at 140 atmospheres rupture pressure, then the initial radius size rc for an adiabatic expansion at k = 7 would have to have been rc = 10-8/ 1.25 = 8 x 10-9 m; moreover, an input of energy sufficient to bring a volume 4/3 π (8 x 10-8)3 to the critical “gas-like” state must have been supplied to the liquid to bring about the rupture. Any initial excited volume smaller than that may indeed form a tiny gas bubble but will immediately thereafter collapse because it is below the critical size required.
4. It may be noted that incipient bubbles, smaller than
those having sufficient excited volume to become critical and bring about macro
rupture of the liquid, may still cause
important transient rupture effects on
the molecular scale. These, while never
reaching the critical radius leading to
macro liquid rupture, may still be of
great importance on the molecular scale in locally removing a water film
barrier between chemical reactant molecules in solution or suspension. This
solvent film barrier phenomenon may therefore also be important in the
kinetics of so-called “slow”
chemical reactions in solution.
Solutions, Solids,
Reaction Kinetics
In simple cases, the relationship of k to n, the number of ways the energy of the system is divided, is given by k = (n +2)/n. With k = 7, the formula would require n to be fractional at n = 1/3, and we would have to then interpret this physically as indicative of the spherical or radial expansion.
For solids, because of structural and steric hindrance, the flow orientation in a rupture flow may conceivably be only quasi- radial, and so a value of k between 4 and 6 might. then be appropriate, giving tensile strengths higher than for liquids but below the classical theoretical estimates. It t would appear that the new model may be of interest to materials science.
Again, the “slow” chemical reactions mentioned in Note 4 above, occur more often in liquid solution than in gases, and they are also the most sensitive to pressure, just as is the case with liquid rupture; furthermore, the reaction rates are slowest when water is the solvent ( Laidler, 1965). This all suggests that the phenomenon of rupture in liquid water may be important in chemical reaction kinetics. In gases, of course, adsorbed molecular films can also be present, and their removal in collision reactions would enter in the same general way as for chemical reaction rates in solution.
Finally, we may note that all the chemical and genetic reactions of life take place in the aqueous medium of the cell. Therefore, the kinetics and probabilities of the reactions of life and its evolution should be subject to the probability laws that govern the aqueous rupture barrier which must be overcome on the molecular scale if the various biochemical reactions and interactions of life are to proceed.
References
Brennen, Christopher E. (1995) Cavitation and Bubble Dynamics. Oxford Univ. Press.
Courant, R. and Friedrichs, K. O. (1948). Supersonic Flow and Shock Waves. Interscience, New York.
Frenkel, J. (1955). Kinetic Theory of Liquids. Dover, New York.
Kittell, Charles. (1968) Introduction to Solid State Physics. , 6th.
ed. John Wiley & Sons Inc.,
Laidler, Keith, J., (1965). Chemical Kinetics. McGraw-Hill.
4.4 Chemical reactions in the aqueous environment of
the Living Cell: An Entropy of Liquid Rupture
Following the above insight into
the rupture of liquids we return to the living cell. It is well known that many
chemical reactions especially those in solution do not take place at the
theoretically predicted rates, but instead take place at rates that are much too slow [29]. For example, there are many
bimolecular reactions (both in gas phase and in solution) which are too slow by
factors up to 10-9. ,so that the rate equation K = Z e –Ea/RT
, (where Z is the collision number, Ea is the activation
energy) must be written as ka
= PZ e –Ea/RT where P the probability
factor is inserted to account for the disparity in reaction rate,
and ka is here the
activation factor or rate [29].
When the collision theory is
replaced by the statistical mechanics approach, the same result is obtained,
but the arbitrary probability factor P is now explained as a steric hindrance
factor which makes more theoretical sense. And since the logarithm of the
probability is the Boltzmann definition of entropy ( d lnP = ∆S) , the
rate equation then takes the form
Krate = PZ e –Ea/RT, and where PZ becomes
PZ = e∆S/R
(kbT/h)
and the entropy of
activation ∆S now appears., with kb
being the Boltzmann constant.
The entropy of activation in
chemical reaction kinetics can easily be related to the new
entropy of rupture ∆SR , in the following manner:
The pressure relationships
across a non-isentropic discontinuity in a compressible fluid may be expressed
as
p1/p2 = e-∆S/R
where ∆S is the entropy change
across it.
Since in any adiabatic process, we also have p1/p2
= (V2/V1)k = (ρ1/ ρ2)k
, therefore we also have p1/p2
= (V2/V1)k = (ρ1/ ρ2)k
= e-∆S/R; and
finally
ln(ρ1/ ρ2)k)
= k ln (ρ1/ ρ2 ) = − ∆S/R
Therefore, ∆S is proportional to k,
and for spherical expansion of water,
k = 7, so ∆S for this case is also proportional to 7.
That is, ∆S![]()
![]() 7.
7.
There
are three main approaches to the relationship of entropy S to probability P. First, in classical thermodynamics,
as in the example above where P = e-∆S/R
, we have ln P =
− ∆S/R. Here P is physically and mathematically equal to
or less than unity.
The
second relationship is the
thermodynamic probability of
Boltzmann and of statistical mechanics where the entropy S is related to the
logarithm of the number of accessible thermodynamic states of the system,
called the thermodynamic probability P or W, which is a number greater than
unity, but which can be
related to the mathematical
probability by a normalization if desired.
Here, ∆S = ln P ( or S = ln W).
Relating this to water rupture, we then have:
(a) From classical chemical
kinetic theory, ln P1 = − ∆S1, where P = e−∆S and so for rupture reactions in
water ln PN = − ∆S ![]() 7 , or log7
P = ln P / ln 7= − ∆S.
7 , or log7
P = ln P / ln 7= − ∆S.
(b) From Boltzman
theory, ln N = − ∆S , where N is the number of accessible states and so for reactions involving water rupture P and N are
related as log 7 P= 7N
(c) A third entropy relationship is from
information theory, where P = loga N, with P now
being the number of bits of information. Consequently, for a system composed of
N assemblages of a bits of thermodynamic information, we have
logaN = P and for
a = 7 this becomes log7 PN =
7N.
If, finally, we make the
assumption that in any time series of evolutionary rupture/ collision events
(i.e. sequential assemblages each having 7 bits of thermodynamic information or
entropy) the probability P of a number
of accessible states is proportional to
the time t available for the process, then
we can put
log7 t = log7P and t = 7N .
When P is normalized to unity this becomes
t = 7N + const.
Therefore,
since evolutionary change takes place by gene reaction in
aqueous solution or suspension in the living cell, and if rupture of
water film is involved in all such
genetic interactions and mutations, and
if water rupture involves an entropy change proportional to 7 and a probability relationship of log P ![]() log t = log7N , then the time series of evolutionary events may also show evidence of a
frequency/probability or entropy
involving the factor 7N
where N is the number of collision/rupture events leading up to the emergence
of the biological form.
log t = log7N , then the time series of evolutionary events may also show evidence of a
frequency/probability or entropy
involving the factor 7N
where N is the number of collision/rupture events leading up to the emergence
of the biological form.
This possibility has been
investigated. In A Logarithmic Scaling of the Time Series of
Cosmic Evolutionary Events to the Base Seven the results do show
a 7N factor. Therefore,
this physically based numerical factor in evolutionary data would seem to merit further
investigation. For convenient reference
some data from the study are as follows:
Notes
1. The times of the events in years ( Nyrs)
are made dimensionless as required for logarithmically
analysis by dividing each time in
years by the first year in the series (No
)which is taken as being unity,
i.e. as year one. Thus N/No
is dimensionless.
5. Plot of Log 7 Data
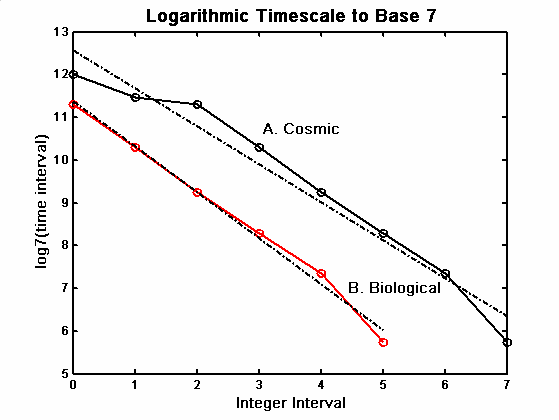
Figure 1. Logarithmic plot to base 7 of (A) all cosmic
data, and (B) biological data only
For Curve A the least squares regression equation is:
(a) negative slope: y = − 0.994 x + 12.18 (b) positive slope: y = + 0.994 x + 5.22 (c) correlation coefficient: 0.998
For curve B the least squares regression equation is:
(a) negative slope: y = − 1.02 x + 11.30 (b) positive slope: y = + 1.02 x + 5.17 (c) correlation coefficient: 0.9988
This empirical
evidence for the presence of a 7N factor in emerging complexity and
evolution is of course preliminary, but
more examples can be found. However, considerably more effort will be needed before it can be said to be
fully established and its scientific implications understood.
4.5 Entropy increase, the 2nd
Law of thermodynamics and evolution.
We have
repeatedly mentioned the problem presented
to all theories of evolution by the 2nd law of thermodynamics, which states that in
natural processes entropy must always increase i.e.
dS = + dQ/dt. Since the emergence
of biological order in the living cell
represents a clear decrease in entropy, this has always been a problem to
explain. Brooks and Wiley [ 1 ] have
endeavored to account for this
contradiction or barrier by postulating
that, while total entropy does always
increase, still, locally, for example in a living cell, the entropy may decreases if the necessary increase in entropy somehow gets exported to the cell ‘s exterior
environment. Although this approach has
not met with general acceptance, it does go directly to the heart of the problem.
We may summarize
our ideas on the situation for material systems as follows:
It appears that the entropy
change laws in the UF in Quadrant I of the p-v thermodynamic diagram should
enter into the basic nature and direction of
plant and animal evolution as
follows:
(a)
Tangent Gas: 1. non-isentropic
processes dS = dQ/T is positive ( 2nd law of thermodynamics holds ( “disorder seeking”)
2. isentropic stable waves : dS = 0
b) Orthogonal ( Isothermal) Gas: 1.
non-isentropic processes dS = dQ/T =
0 ( “stability
seeking” )
2.
isentropic stable waves dS = dQ/T = 0
The detailed interaction of
ordinary physical processes with the
tangent gas and the isothermal gas entropy laws of the UF where the forces
originate, remains to be examined.
The existence of stable waves in the UF provides for the orderly
forces observed in physical processes and supplies the observed order which
emerges in complexity.
The existence of a quite
separate “stability seeking” entropy in the UF’s isothermal gas raises the
question of whether the emergence of biological complexity might not then be
essentially different in nature from emerging complexity of non-living physical
systems. If this is so, then, not only the existence of orderly forces must be
considered, but also the existence of an essentially different entropy system ,
one that is intrinsically not only affected by order but actually seeks the
stability of order instead of merely tolerating it. Physical complexity would
then arise from the forces of the UF acting under the 2nd law. But
living plant and animals would arise from the same orderly forces, plus the
‘stability- seeking’ flow of entropy in the isothermal UF gas. The emergence of
complexity in biological systems would then have an essentially different
ingredient from its emergence in non-living physical systems.
[Note: There is another entropy condition in the UF , namely that
in Quadrant II (dS = − dQ/T) ;
this implies very unusual
properties which appear to match the human condition. This is discussed briefly
in Section 5 below, and in detail in Science and the
Soul/Body Problem: An Exploratory Reassessment .
It is now time to
sum up this survey.
 5.0
Conclusions on Biological Complexity: Do Orderly Forces Acting on Random
Kinetic Motions Resuslt in the Emergence of Variety on a Probabilistic Time
Scale?
5.0
Conclusions on Biological Complexity: Do Orderly Forces Acting on Random
Kinetic Motions Resuslt in the Emergence of Variety on a Probabilistic Time
Scale?
We
started by examining the proposition
that evolution, defined as the emergence of complexity, both physical and
biological, arises in chaotic or kinetic
systems which, when not at equilibrium,
receive an input of “stressing”
energy. The only elements considered in this system are suitable material particles ( essentially molecules
and gene pools) undergoing energetic, random (i.e. kinetic) motions.
Our
analysis concluded that several physical elements were thereby ignored and must
be included in any valid explanatory system. These include, the presence
of (1) orderly forces ( which we have
argued are best explained by the existence of a universal wave and force field
(UF) (2) acting on random kinetic
motions of molecular species in space and
over time, and (3) in the case of
the aqueous environment of the living cell, also involving the entropy change in the rupture of water barriers between
reacting molecular assemblages, and (4)
the presence of two new orders for entropy change direction governing force interactions with the UF, in
addition to the long standing rule of the
2nd law of thermodynamics for kinetic processes among
molecular species.
If
the orderly forces are, say, a UF wave passing through a kinetic motion field,
then, because the wave is extended in space it will affect the kinetic field at
one point only over a certain interval
of time set by the wave speed. This will
inevitably introduce further
quasi-ordered variation into the kinetic field over the space of the
affected field and over the elapsed time at any one point in the field. The order
of the UF wave is therefore not manifested in the physical field
directly, but instead appears in a space- modified and time- dependent manner at any one location.
This would seem to mean the emergence of variety
from the interaction of UF wave order
and random physical kinetic field. The process is deterministic in that the UF imposes order, but it is probabilistic in the time
sequence in which the realized ordered
system emerges and in the variety of the realized order that
interaction makes possible. The
various physical complexity examples discussed by Nicolis and Prigogine could
be re-analysed from this point of view; e.g.
snowflakes emerge from a super
cooled cloud under the action of an orderly vapour force field in orderly
types, but they emerge individually on a probabilistic time scale and are
individually highly non- uniform in their fine scale structure.
Four
obvious approaches to a better understanding of this new scheme would seem to be : (1) a re-analysis of the
emergence of physical complexity with the inclusion of orderly physical force fields, and (2) a detailed quantitative
analysis of molecular genetic and metabolic
interactions in the aqueous cell medium,
(3) a theoretical analysis of the interaction of order and probability
in physical systems, (4) evaluation of the implications for biology of the two
new orders of entropy change now theoretically available from the UF (namely
“order stability” and “order seeking”).
The
suggested new approaches, if eventually validated, may not greatly affect
much of current biological
investigation, working theories,
and progress, for example in the
marvelous structural complexities being revealed by molecular biology. However, a new attention to the force fields
involved in these processes should open up many new avenues for experimental
exploration and verification. At the theoretical or conceptual level, it should perhaps also be noted that the
interpretation of the conclusions
extends well beyond the autonomy and methods of science into the other two methods of the rational investigation of reality, namely
philosophy ( ontology, being ) and theology ( ultimate meaning).
In
summary:
1.The
neo-Darwinian proposition that biological evolution arises out of purely random
physical processes alone, and then proceeds via probability laws, is untenable,
since it ignores the orderly
physically forces that are
acting.
2.
The proposition that evolution arises from a combination of kinetic disorder
plus orderly forces is presently tenable for non-living physical systems, but
it still does not account for the violation of the 2nd law in the emerging
complexity of biological systems.
3. The proposition that the
UF is the source of physical force ( wave energy exchange ), structural and
flow order (entropy minimization) is tenable, and therefore the UF’s entropy change relationships must also be
acting in any interaction with atomic and molecular material systems. These
relationships appear now to a) agree
with the 2nd Law for the tangent gas in Quadrant I of the
pressure-volume diagram, b) require zero entropy change in material interactions with the UF’s orthogonal or
isothermal gas, c) require negative
entropy change in interactions with the UF’s dynamic entity in Quadrant II.
4. In addition to the
presence of orderly forces, there are now three entropy change regimes,
instead of only the 2nd law, that must
be considered in evolutionary change.
5. These new entropy orders
or regimes appear to point towards an essential
entropic difference between non-living and living systems in the case of the UF’s orthogonal/isothermal
dynamic order in Quadrant I, where
non-living physical systems obey the 2nd Law and living
systems obey the UF’s orthogonal/isothermal gas law.
6. In the case of the UF
in Quadrant II, the entropy decrease law (dS = −dQ/ T) appears to point towards an essential difference between plant/animal systems on
the one hand and human beings on the other.
If this property of the UF is valid as interpreted here, then science
may be at a frontier between itself and philosophy/ theology. Science is
ordinarily thought of as being concerned with measurement and with the
quantitative laws of the behaviour of matter. However, Quadrant II in the UF
appears to present a dynamic, non-material substance, extrinsically rather than
intrinsically quantified, and “ order seeking” in its entropy law, i.e tending towards the intellectual.
`
The new statement of entropy
change appears to be theoretically
valid, although it may not be directly experimentally verifiable (or
falsifiable). In short : the differential equation dS = + dQ/T is a rational, mathematical, scientific,
physical statement and a verified scientific
law describing the direction of flow of non-biological ( the 2nd Law of
thermodynamics) . What then is the nature of the statement made by the
differential equation dS = −dQ/T , both
physically and metaphysically? It
also is rational, mathematically sound, makes a physical statement, and
describes the flow of information and order seeking, or insight, in human intellectual capabilities.
Does it also raise a question
as to whether the statement constitutes a frontier between, or a bridge to,
philosophy/ theology, which have
rationally affirmed the reality of spirit for millennia?
It might then be tentatively
restated, and rather loosely, that the new theory conceptually means that (1)
the quantitative interaction of the UF
with material particles (condensed from it in Quadrant I [23]) and the
subsequent cosmic and biological evolution is a concern of science, (2) the interaction of the UF in Quadrant II ( soul/spirit) with matter in
Quadrant I constitutes human life whose evolution is a subject of prehistory and then
history, (3) the meaning of the theory
is a concern of philosophy, and (4) the origin of the UF, its purpose and
destiny, and the purpose and destiny of humanity are the concerns of both
philosophy and religion.
7. In the mid-1950’s, Lonergan [30] critically
analyzed a wide variety of scientific theories as instances of human insight (
and oversight) and of the nature of
rationality or the act of human understanding which leads to the development of
natural science, philosophy and theology. He then developed a philosophic world
view which took into account both the
classical deterministic physical laws
and the statistical probabilities of experimental science in a synthesis which he termed emergent probability. It might
be of interest to now see how his emergent probability alters or
survives when the UF and its laws are included.
The new orderly wave and force field would appear to relate to his
classical deterministic laws so as to make them more concrete, while the new
entropy laws would appear to introduce
discontinuities which separate distinct orders of existence, and which must also fundamentally affect
probabilities attached to the emergence of events.
References
1. Brooks, Daniel R., and E.O. Wiley. Evolution
as Entropy. University of Chicago Press, Chicago and London, 1986.
2. Nicolis, Gregoire and Ilya
Prigogine. Exploring Complexity, W. H. Freeman and Company, New York, 1989.
3. S. A. Chaplygin, On Gas Jets.
Sci. Mem. Moscow Univ. Math. Phys. 21,
1 (1904).
4. H. S. Tsien,
Two-Dimensional Subsonic Flow of Compressible Fluids. J.
Aeron. Sci. 6, 399 (1939).
5. T. Von Karman,
Compressibility Effects in Aerodynamics.
J. Aeron. Sci. 8, 337 (1941).
6. A. H. Shapiro, The Dynamics and Thermodynamics of
Compressible Fluid Flow. 2 Vols.( John Wiley & Sons, New York, 1953).
7. R. Courant and K. O. Friedrichs, Supersonic
Flow and Shock Waves. (Interscience , New York, 1948).
8.
Horace Lamb, Hydrodynamics. 6th ed . ( Dover Reprint, Dover Publications Inc. New York, 1936 ).
9. N. A. Bachall, J.P. Ostriker, S Perlmutter and P.J.
Steinhardt. The Cosmic Triangle:
Revealing the State of the Universe. Science,
284, 1481 (1999).
10. A. Kamenshchick, U. Moschella and V.
Pasquier, An alternative to
quintessence. Phys Lett. B
511, 265 (2001).
11. N. Bilic, G.B. Tupper and
R.D.Viollier, Unification of Dark Matter
and Dark Energy: The Inhomogeneous Chaplygin Gas. Astrophysics, astro-ph/0111325
( 2002).
12.
Power, Bernard A., Much of the work leading up to the present theory has
appeared in connection with studies into
the scientific basis for the image formation on the Holy Shroud of
Turin. Some of these are as follows:
13. ---------------, Il Meccanismo di
Formazione dell’Immagine dela Sindon di Torino, Collegamento pro Sindone, Mgggio-Giugno, pp. 13-28, 1997, Roma.
14.---------------, Caratterizzazione di
una Lunghezza d’Onda per la Radiazione che Potrebe aver Creato I’Immagine Della
Sindone di Torino. Collegamento pro
Sindone, Roma. Novembre-Decembre, pp. 26-36, 1999.
15.---------------, An Unexpected
Consequence of Radiation Theories of Image formation for the Shroud of Turin. Proc. Worldwide Congress Sindone 2000, Orvieto, Italy, Aug. 27-29, 2000.
16.---------------, Image Formation on the
Holy Shroud of Turin by Attenuation of Radiation in Air. Collegamento
pro Sindone website (www.shroud.it/) March 2002.
17.---------------, How Microwave Radiation Could Have
Formed the Observed Images on The Holy Shroud of Turin. Collegamento [ro Sindone Website, Jan. 2003. (www.shroud.it/)
18. --------------, Shock Waves in a Photon
Gas. Contr. Paper No. 203, American
Association for the Advancement of Science, Ann. Meeting, Toronto, Jan.
1981.
19.---------------. Unification of Forces
and Particle Production at an Oblique Radiation Shock Front. Contr. Paper N0. 462. American
Association for the Advancement of
Science, Ann. Meeting, Washington,
D.C., Jan 1982.
20.---------------, Baryon Mass-ratios and
Degrees of Freedom in a Compressible Radiation Flow. Contr.
Paper No. 505. American Association for the Advancement of Science, Annual
Meeting, Detroit, May 1983.
21.---------------,
Summary of a Universal Physics. Monograph (Private distribution) pp 92.
Tempress, Dorval, Quebec, 1992.
22. ……………,
Properties of a Universal Wave Field (UF). http://www.shroudscience.info/
November, 2005.
23. ……………, Cosmology of a Binary
Universe, Part 1: The Origin, Properties and Thermodynamic Evolution of a
Universal Wave and Force Field. http://www.shroudscience.info/.
April 2007.
24. L. W. Gold and B. A. Power. Dependence of the forms of natural
snow crystals on meteorological conditions. J. Meteorology S9, 447 (1952).
25. Nakaya, Ukichiro. Snow Crystals: Natural and Artificial.
Harvard University Press, Cambridge, Mass.
1954.
26. B. A. Power and R.F. Power. Some Amino
Acids as Ice Nucleators. Nature,
Vol. 184, 1170. 1962.
27.
-------------------------------------
Vanillin, cis- Terpin and cis- Terpin Hydrate as Ice Nucleators. Science.
Vol. 148, 1088, 21 May, 1965.
28.
Brennen, Christopher, E. Cavitation
and Bubble Dynamics. Oxford Univ. Press. 1995.
29. Laidler,
Keith, J., Chemical Kinetics. McGraw-Hill. 1965.
30. Lonergan,
Bernard , S.J., Insight: A Study of Human
Understanding. Philosophical
Library, New York. 1956.
Copyright January 2008 Bernard A Power..\index.htm